Volume 9, Issue 3 (7-2023)
Journal of Research in Applied and Basic Medical Sciences 2023, 9(3): 176-182 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Sheikhi E, Aghamaali M, Babataheri S. Investigating the combined effect of ursolic acid and alpelisib on inhibiting cell proliferation and the expression of HIF1α on 4T1 breast cancer cell line. Journal of Research in Applied and Basic Medical Sciences 2023; 9 (3) :176-182
URL: http://ijrabms.umsu.ac.ir/article-1-273-en.html
URL: http://ijrabms.umsu.ac.ir/article-1-273-en.html
Department of Biochemistry, Faculty of Basic Sciences. University of Guilan, Rasht, Iran , aghamaali@guilan.ac.ir
Full-Text [PDF 303 kb]
(545 Downloads)
| Abstract (HTML) (1730 Views)
Table 1. Characteristics of primers used in Real-Time PCR
Table 2. Device program for Real-Time PCR reaction
The fold change of HIF1α gene expression in different groups was calculated using the 2-ΔΔCT formula.
Statistical analysis:
Statistical analysis performed using GraphPad Prism version 8.4 software. Values are shown as means ± S.D. One-way ANOVA was performed to determine the significance between groups. Confidence limits for all tests were considered 95% and P < 0.05 were considered to indicate a significant difference.
Results
The IC50 values of Ursolic acid and Alpelisib were 168.314 μM and 6.377 μM, respectively.
It was observed that 168.314 µM Ursolic acid and 6.377 µM Alpelisib markedly suppressed breast cancer cell viability in 4T1 cell line in a concentration-dependent manner (P<0.05).
Based on these results, all subsequent experiments were performed using IC50 concentration for single treatment and ½ IC50 for combination groups. The results of cell viability based on MTT assay are shown in Figure 1.

Fig. 1. Shows an anti-proliferative effects of Ursolic acid and Alpelisib on the 4T1 cells. The inhibitory effects of Ursolic acid and Alpelisib with different concentrations after 24 h incubation. Results are expressed as percent of cell proliferation of control at 0 h. The data shown are the mean from three independent experiments, each with triplicate wells. The result indicates a significant difference between control and single drug-treatment cells as analyzed by Dunnett’s test (P<0.05). The IC50 values of Ursolic acid and Alpelisib were 168.314 μM and 6.377 μM, respectively.
Treatment of cells with IC50 concentration of Alpelisib and Ursolic acid led to a significant decrease in HIF1α gene expression compared to the control group. The decrease in the expression of HIF1α in the single-treatment and combination-treatment groups was statistically significant, compared to the control group. These results probably indicate that the effect of these drugs on viability and growth of cancer cells by directing these cells towards apoptosis by regulating the genes involved in the PI3K/AKT/mTOR signaling pathway.
Hypoxia occurs in most solid tumors, and has been associated not only with malignant progression and poor prognosis but also with specific resistance to anti-cancer therapies. In normal cells, the level of HIF1α is not much enough to measure accurately. Therefore, increasing its expression indicates that HIF1α plays a critical role in tumor progression and expansion (3, 9).
Biochemical analyzes have shown that tumor suppressor p53 could directly interact with HIF1α. Thus, HIF1α stabilizes p53 and induces apoptosis in hypoxia conditions. On the other hand, it has been shown that HIF1α has a stronger role than P53 in inducing cell cycle arrest (10). Alpelisib is FDA approved, orally active, and highly selective inhibitor of the phosphatidylinositol 3-kinase (PI3K) signaling pathway. This pathway is known to regulate the expression of hypoxia-inducible factor-1 alpha (HIF1α), which plays a crucial role in the cellular response to hypoxia or low oxygen levels. HIF1α is involved in various cellular processes, such as angiogenesis, erythropoiesis, glucose metabolism, and many others (6, 11).
On the other hand, Ursolic acid is a natural pentacyclic triterpenoid that has been shown to have various pharmacological properties. Recent studies have revealed that Ursolic acid can suppress the PI3K/Akt signaling pathway, which is downstream of PI3K and further contributes to the regulation of HIF1alpha (8, 12).
In previous studies, HT-29 colorectal carcinoma cell line was treated with a combination of Ursolic acid and oxaliplatin; the combination of drugs significantly inhibited p-AKT. Cells treated with the combination of drugs showed the significant tumor inhibition, indicating the potential of Ursolic acid to exert a synergistic effect in combination with other drugs (13).
Several research studies have suggested that both Alpelisib and Ursolic acid can modulate the expression of HIF1α, either directly or indirectly. For instance, a study by Kuroda et al. (2020) reported that treatment with Alpelisib reduced the expression of HIF1α in triple-negative breast cancer cells, which are known to be highly dependent on PI3K/mTOR signaling for their growth and survival. The authors suggested that Alpelisib could be a promising therapeutic option for these types of cancers by targeting the HIF1α pathway (14).
Similarly, other studies demonstrated that Ursolic acid treatment inhibited the expression of HIF1α and downstream targets in prostate cancer and hepatocellular carcinoma cells. In addition, different classes of terpenoids produce a synergistic effect when combined with standard chemotherapeutic agents or targeted agents. The researchers proposed that Ursolic acid could exert its anti-tumor effects by disrupting the PI3K/Akt/HIF1 alpha axis, which plays a crucial role in tumorigenesis and angiogenesis (15). The inhibitory effect of Alpelisib and Ursolic acid on different cell lines has also been studied. The inhibitory effect of Ursolic acid on MCF7 cell line has been shown by researchers. In addition, the induction of apoptosis of HT-29 cells by Ursolic acid has been shown in the study of Chen et al. Chteinberg et al. have shown the effect of Ursolic acid alone and in combination with Navitoclax on the induction of apoptosis in MCC cell line. Suppression of HIF1α gene by Ursolic acid in other cell lines such as Hep3B، Huh7, RKO، LoVo, SW480, and HA22T has also been shown in other studies (16). Ursolic acid and Alpelisib has been used in separately with other drugs and in some cases, it has shown synergistic effects. Based on previous researches, the combination of Alpelisib with MCL1 inhibitor (s63845) and Fulvestrant have significant synergistic effects in inhibiting the growth of cancer cells (8, 17-19).
In the current study, we hypothesized that UA induced its anti-tumor effect by regulating the expression of HIF1α. Real-time PCR analysis confirmed that HIF1α mRNA expression was decreased in both single-treatment and mix-treatment groups in 4T1 cells. This finding reveals that Ursolic acid and Alpelisib down regulate the expression of the HIF1α in 4T1 cells, leading to inhibition of PI3K/Akt axis and subsequent apoptosis induction via regulation of downstream elements.
Taken together, both Ursolic acid and Alpelisib, demonstrated strong effect on viability of 4T1 breast cancer cell line and gene expression on HIF1α. These findings suggest that relevant compounds could be potential therapeutic options for hypoxia-related diseases and cancers that are regulated by the HIF1α pathway. Further studies are required to establish the mechanism of reactions between related drugs and other pathways. Furthermore, these findings should be evaluated with in vivo animal models, which can facilitate and accredit the in vitro results.
Acknowledgments
No Declared
Conflict of interest
No conflict of interest declaration between the authors.
Funding/support
No funding source.
Full-Text: (638 Views)
Introduction
Breast cancer is the most prevalent leading cause of mortality associated with cancer among females worldwide. Like most cancers, Breast cancer is clinically and molecularly a heterogeneous disease. Tumor types and molecular profile characteristics play an important role in choosing therapeutic strategies. Effective treatment of breast cancer
requires a multidisciplinary approach comprising surgery, radiotherapy, adjuvant and neoadjuvant therapy (1). Breast cancer is classified into five subtypes: luminal-A, luminal-B, normal-like, basal-like, and HER2-enriched (2). Mutations in specific genes such as HIF1α, P53,CDKs etc probably increase the risk of breast cancer more than other genes (1).
Hypoxia is a feature in most of the solid tumors and is associated with poor prognosis in several cancer types, including breast cancer. The master regulator of the hypoxic response is the Hypoxia-inducible factor 1α (HIF1α). The basis for the association between hypoxia, tumorigenesis and malignant progression has been examined extensively at both the molecular and the cellular level. Hypoxia exerts direct effects on the expression of numerous gene products involved in processes such as angiogenesis, apoptosis, glycolysis and cell-cycle control, which are central to the survival and expansion of a malignant cell population in an oxygen-deficient circumstance (3). HIF1α is a transcription factor that plays a key role in regulating the survival and growth of tumor cells in hypoxic environments. Therefore, inhibition of HIF1α expression is an important target in cancer therapy (4).
On the other hand, Alpelisib as an FDA approved anti-cancer drug is used in combination with Fulvestrant for the treatment of metastatic breast cancer. Alpelisib belongs to the group of kinase inhibitors that are used to inhibit the overgrowth related to the PIK3CA and cancer cell multiplication. Studies have shown that Alpelisib can down-regulate HIF1α gene expression in breast cancer cells. This effect may be due to the inhibition of the PI3K/Akt/ mTOR signaling pathway, which is known to activate HIF1α gene expression (5, 6).
Ursolic acid is a natural pentacyclic triterpene compound found in a variety of plants which shows many beneficial effects such as anti-inflammatory, antioxidant, anti-apoptotic, and anti-carcinogenic effects. It exerts anti-carcinogenic effects by suppressing nuclear factor-kappa β (NFkβ) signaling and increasing the antioxidants level. Ursolic acid is used as an alternative treatment for cancer in this study. Ursolic acid and Alpelisib are two compounds that have been shown to have effects on gene expression, specifically on the HIF1α gene. Some studies have shown that Ursolic acid inhibits HIF1α expression in human hepatocellular carcinoma cells. HIF1α plays a crucial role in regulating the body's response to hypoxia, or low oxygen levels, and is involved in a number of important cellular processes, including metabolism, cell growth, and differentiation (7, 8).
In the present study, it was hypothesized that Ursolic acid and Alpelisib may inhibit the proliferation of breast cancer cells in vitro, and may induce apoptosis via down regulation of HIF1α to suppress the development of breast cancer.
Materials & Methods
Cell line and culture conditions:
4T1 cell line was purchased from Pasteur Institute of Iran. Cells were cultured in 75 flasks using RPMI 1640 medium (Gibco, USA) containing 10% FBS (Fetal Bovine Serum) (Biosera, France) and 1% (Penicillin-Streptomycin) (Gibco, USA) under conditions of 5% CO2, humidity 90% and 37°C. The culture medium was changed three times a week and trypsin/EDTA solution was used to harvest the cells. Then the third passage cells were used for the next steps.
Medicines and their preparation and used concentrations:
Ursolic acid and Alpelisib were prepared from SIGMA-ALDRICH and MEDCHEM EXPRESS companies, respectively. The solvents used were 99% ethanol, PBS and DMSO. Cell cultures were divided into two test groups including mono-drug and dual-drug groups. In the single-drug group, concentrations of 25, 50, 100, 200, and 400 μM of Ursolic acid and 0.5, 1, 2, 4, and 8 μM of Alpelisib were used. Three groups without any additional solution, Ursolic acid solvent, and Alpelisib solvent were used as control groups.
Determination of effective dose:
The effective dose of Ursolic acid and Alpelisib was analyzed by MTT assay. The MTT test was repeated three times, and the average absorbance obtained from three repetitions of the MTT test was included in the vital power formula. The IC50 value of each drug was determined using Compusyn software, and the corresponding graph was drawn with GraphPad Prism software.
Real-time PCR:
Concentrations equal to IC50 for single-drug groups and half of IC50 concentration for two-drug groups were used to investigate gene expression by Real-time PCR method. 3×106 cells were used in the culture medium for each group. After incubation for 24 hours, cells were washed with PBS buffer, and total RNA was extracted by Hybrid-R kit (GeneAll, South Korea) according to the company's instructions. cDNA synthesis was performed using a commercial kit (YektaTajhiz, Iran) according to the manufacturer's protocol. The Real-time PCR reaction was performed using the primers for GAPDH as housekeeping gene and HIF1α as the target gene given in table 1 and according to the information given in table 2.
Breast cancer is the most prevalent leading cause of mortality associated with cancer among females worldwide. Like most cancers, Breast cancer is clinically and molecularly a heterogeneous disease. Tumor types and molecular profile characteristics play an important role in choosing therapeutic strategies. Effective treatment of breast cancer
requires a multidisciplinary approach comprising surgery, radiotherapy, adjuvant and neoadjuvant therapy (1). Breast cancer is classified into five subtypes: luminal-A, luminal-B, normal-like, basal-like, and HER2-enriched (2). Mutations in specific genes such as HIF1α, P53,CDKs etc probably increase the risk of breast cancer more than other genes (1).
Hypoxia is a feature in most of the solid tumors and is associated with poor prognosis in several cancer types, including breast cancer. The master regulator of the hypoxic response is the Hypoxia-inducible factor 1α (HIF1α). The basis for the association between hypoxia, tumorigenesis and malignant progression has been examined extensively at both the molecular and the cellular level. Hypoxia exerts direct effects on the expression of numerous gene products involved in processes such as angiogenesis, apoptosis, glycolysis and cell-cycle control, which are central to the survival and expansion of a malignant cell population in an oxygen-deficient circumstance (3). HIF1α is a transcription factor that plays a key role in regulating the survival and growth of tumor cells in hypoxic environments. Therefore, inhibition of HIF1α expression is an important target in cancer therapy (4).
On the other hand, Alpelisib as an FDA approved anti-cancer drug is used in combination with Fulvestrant for the treatment of metastatic breast cancer. Alpelisib belongs to the group of kinase inhibitors that are used to inhibit the overgrowth related to the PIK3CA and cancer cell multiplication. Studies have shown that Alpelisib can down-regulate HIF1α gene expression in breast cancer cells. This effect may be due to the inhibition of the PI3K/Akt/ mTOR signaling pathway, which is known to activate HIF1α gene expression (5, 6).
Ursolic acid is a natural pentacyclic triterpene compound found in a variety of plants which shows many beneficial effects such as anti-inflammatory, antioxidant, anti-apoptotic, and anti-carcinogenic effects. It exerts anti-carcinogenic effects by suppressing nuclear factor-kappa β (NFkβ) signaling and increasing the antioxidants level. Ursolic acid is used as an alternative treatment for cancer in this study. Ursolic acid and Alpelisib are two compounds that have been shown to have effects on gene expression, specifically on the HIF1α gene. Some studies have shown that Ursolic acid inhibits HIF1α expression in human hepatocellular carcinoma cells. HIF1α plays a crucial role in regulating the body's response to hypoxia, or low oxygen levels, and is involved in a number of important cellular processes, including metabolism, cell growth, and differentiation (7, 8).
In the present study, it was hypothesized that Ursolic acid and Alpelisib may inhibit the proliferation of breast cancer cells in vitro, and may induce apoptosis via down regulation of HIF1α to suppress the development of breast cancer.
Materials & Methods
Cell line and culture conditions:
4T1 cell line was purchased from Pasteur Institute of Iran. Cells were cultured in 75 flasks using RPMI 1640 medium (Gibco, USA) containing 10% FBS (Fetal Bovine Serum) (Biosera, France) and 1% (Penicillin-Streptomycin) (Gibco, USA) under conditions of 5% CO2, humidity 90% and 37°C. The culture medium was changed three times a week and trypsin/EDTA solution was used to harvest the cells. Then the third passage cells were used for the next steps.
Medicines and their preparation and used concentrations:
Ursolic acid and Alpelisib were prepared from SIGMA-ALDRICH and MEDCHEM EXPRESS companies, respectively. The solvents used were 99% ethanol, PBS and DMSO. Cell cultures were divided into two test groups including mono-drug and dual-drug groups. In the single-drug group, concentrations of 25, 50, 100, 200, and 400 μM of Ursolic acid and 0.5, 1, 2, 4, and 8 μM of Alpelisib were used. Three groups without any additional solution, Ursolic acid solvent, and Alpelisib solvent were used as control groups.
Determination of effective dose:
The effective dose of Ursolic acid and Alpelisib was analyzed by MTT assay. The MTT test was repeated three times, and the average absorbance obtained from three repetitions of the MTT test was included in the vital power formula. The IC50 value of each drug was determined using Compusyn software, and the corresponding graph was drawn with GraphPad Prism software.
Real-time PCR:
Concentrations equal to IC50 for single-drug groups and half of IC50 concentration for two-drug groups were used to investigate gene expression by Real-time PCR method. 3×106 cells were used in the culture medium for each group. After incubation for 24 hours, cells were washed with PBS buffer, and total RNA was extracted by Hybrid-R kit (GeneAll, South Korea) according to the company's instructions. cDNA synthesis was performed using a commercial kit (YektaTajhiz, Iran) according to the manufacturer's protocol. The Real-time PCR reaction was performed using the primers for GAPDH as housekeeping gene and HIF1α as the target gene given in table 1 and according to the information given in table 2.
Table 1. Characteristics of primers used in Real-Time PCR
| Gene | Forward primer sequence | Reverse primer sequence | Primer length | Temperature (̊c) |
| GAPDH | CAACTCCCACTCTTCCACCT | GAGTTGGGATAGGGCCTCTC | 205 | 60 |
| HIF1α | TCAGCATACAGTGGCACTCA | AAGGGAGCCATCATGTTCCA | 213 | 60 |
Table 2. Device program for Real-Time PCR reaction
| Step | Time | Temperature (◦C) | Cycle | |
| Primary denaturation | 10 | minutes | 94 | 1 |
| DENATURATION | 30” | 94 | 40 | |
| Annealing | 60” | 60 | ||
| elongation | 60” | 72 | ||
| final elongation | 10” | 72 | 1 | |
The fold change of HIF1α gene expression in different groups was calculated using the 2-ΔΔCT formula.
Statistical analysis:
Statistical analysis performed using GraphPad Prism version 8.4 software. Values are shown as means ± S.D. One-way ANOVA was performed to determine the significance between groups. Confidence limits for all tests were considered 95% and P < 0.05 were considered to indicate a significant difference.
Results
The IC50 values of Ursolic acid and Alpelisib were 168.314 μM and 6.377 μM, respectively.
It was observed that 168.314 µM Ursolic acid and 6.377 µM Alpelisib markedly suppressed breast cancer cell viability in 4T1 cell line in a concentration-dependent manner (P<0.05).
Based on these results, all subsequent experiments were performed using IC50 concentration for single treatment and ½ IC50 for combination groups. The results of cell viability based on MTT assay are shown in Figure 1.

Fig. 1. Shows an anti-proliferative effects of Ursolic acid and Alpelisib on the 4T1 cells. The inhibitory effects of Ursolic acid and Alpelisib with different concentrations after 24 h incubation. Results are expressed as percent of cell proliferation of control at 0 h. The data shown are the mean from three independent experiments, each with triplicate wells. The result indicates a significant difference between control and single drug-treatment cells as analyzed by Dunnett’s test (P<0.05). The IC50 values of Ursolic acid and Alpelisib were 168.314 μM and 6.377 μM, respectively.
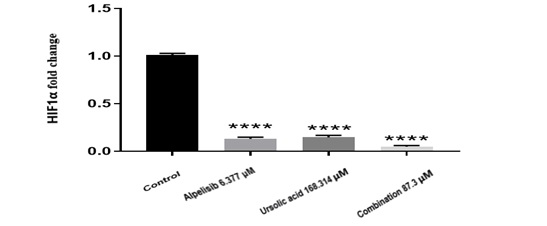
Treatment of cells with IC50 concentration of Alpelisib and Ursolic acid led to a significant decrease in HIF1α gene expression compared to the control group. The decrease in the expression of HIF1α in the single-treatment and combination-treatment groups was statistically significant, compared to the control group. These results probably indicate that the effect of these drugs on viability and growth of cancer cells by directing these cells towards apoptosis by regulating the genes involved in the PI3K/AKT/mTOR signaling pathway.

Fig. 2. Expression of HIF1α in single treatment and combination of drugs on 4T1 cells. Cells were cultured with 168.314 μM UA and 6.377 ALP for 24-h, and total RNA was isolated. Real-time PCR was performed using SYBR Green I as the detection dye after reverse transcription with Oligo (dT) primers. Ct was measured, and HIF1α expression was presented as 2-∆Ct, with ∆Ct = Ct HIF1α – Ct GAPDH. Compared with vehicle control, HIF1α mRNA was significantly decreased in single-treatment groups and combination groups (*P<0.05). Data are expressed as the mean ± standard error of the mean. Values given were derived from the average of at least three independent experiments. *P<0.05, **P<0.01, ***P<0.001 and, ****P<0.0001 denote means significantly different from untreated cells.
Table 3. The results of One Way ANOVA based on Tukey's test according to alpha coefficient (0.05) for HIF1α gene. Tukey's test is used to compare the mean variances of groups. Also, this test indicates the overall error.
DiscussionTable 3. The results of One Way ANOVA based on Tukey's test according to alpha coefficient (0.05) for HIF1α gene. Tukey's test is used to compare the mean variances of groups. Also, this test indicates the overall error.
| Test details | Mean diff | SE of diff | 95.00% CI of diff | P value |
| Control VS. Alp | 0.8823 | 0.01783 | 0.8177 to 0.9468 | ****<0.0001 |
| Control VS. UA | 0.8663 | 0.01783 | 0.8017 to 0.9308 | ****<0.0001 |
| Control VS. Alp+UA | 0.9632 | 0.01783 | 0.8987 to 1.028 | ****<0.0001 |
Hypoxia occurs in most solid tumors, and has been associated not only with malignant progression and poor prognosis but also with specific resistance to anti-cancer therapies. In normal cells, the level of HIF1α is not much enough to measure accurately. Therefore, increasing its expression indicates that HIF1α plays a critical role in tumor progression and expansion (3, 9).
Biochemical analyzes have shown that tumor suppressor p53 could directly interact with HIF1α. Thus, HIF1α stabilizes p53 and induces apoptosis in hypoxia conditions. On the other hand, it has been shown that HIF1α has a stronger role than P53 in inducing cell cycle arrest (10). Alpelisib is FDA approved, orally active, and highly selective inhibitor of the phosphatidylinositol 3-kinase (PI3K) signaling pathway. This pathway is known to regulate the expression of hypoxia-inducible factor-1 alpha (HIF1α), which plays a crucial role in the cellular response to hypoxia or low oxygen levels. HIF1α is involved in various cellular processes, such as angiogenesis, erythropoiesis, glucose metabolism, and many others (6, 11).
On the other hand, Ursolic acid is a natural pentacyclic triterpenoid that has been shown to have various pharmacological properties. Recent studies have revealed that Ursolic acid can suppress the PI3K/Akt signaling pathway, which is downstream of PI3K and further contributes to the regulation of HIF1alpha (8, 12).
In previous studies, HT-29 colorectal carcinoma cell line was treated with a combination of Ursolic acid and oxaliplatin; the combination of drugs significantly inhibited p-AKT. Cells treated with the combination of drugs showed the significant tumor inhibition, indicating the potential of Ursolic acid to exert a synergistic effect in combination with other drugs (13).
Several research studies have suggested that both Alpelisib and Ursolic acid can modulate the expression of HIF1α, either directly or indirectly. For instance, a study by Kuroda et al. (2020) reported that treatment with Alpelisib reduced the expression of HIF1α in triple-negative breast cancer cells, which are known to be highly dependent on PI3K/mTOR signaling for their growth and survival. The authors suggested that Alpelisib could be a promising therapeutic option for these types of cancers by targeting the HIF1α pathway (14).
Similarly, other studies demonstrated that Ursolic acid treatment inhibited the expression of HIF1α and downstream targets in prostate cancer and hepatocellular carcinoma cells. In addition, different classes of terpenoids produce a synergistic effect when combined with standard chemotherapeutic agents or targeted agents. The researchers proposed that Ursolic acid could exert its anti-tumor effects by disrupting the PI3K/Akt/HIF1 alpha axis, which plays a crucial role in tumorigenesis and angiogenesis (15). The inhibitory effect of Alpelisib and Ursolic acid on different cell lines has also been studied. The inhibitory effect of Ursolic acid on MCF7 cell line has been shown by researchers. In addition, the induction of apoptosis of HT-29 cells by Ursolic acid has been shown in the study of Chen et al. Chteinberg et al. have shown the effect of Ursolic acid alone and in combination with Navitoclax on the induction of apoptosis in MCC cell line. Suppression of HIF1α gene by Ursolic acid in other cell lines such as Hep3B، Huh7, RKO، LoVo, SW480, and HA22T has also been shown in other studies (16). Ursolic acid and Alpelisib has been used in separately with other drugs and in some cases, it has shown synergistic effects. Based on previous researches, the combination of Alpelisib with MCL1 inhibitor (s63845) and Fulvestrant have significant synergistic effects in inhibiting the growth of cancer cells (8, 17-19).
In the current study, we hypothesized that UA induced its anti-tumor effect by regulating the expression of HIF1α. Real-time PCR analysis confirmed that HIF1α mRNA expression was decreased in both single-treatment and mix-treatment groups in 4T1 cells. This finding reveals that Ursolic acid and Alpelisib down regulate the expression of the HIF1α in 4T1 cells, leading to inhibition of PI3K/Akt axis and subsequent apoptosis induction via regulation of downstream elements.
Taken together, both Ursolic acid and Alpelisib, demonstrated strong effect on viability of 4T1 breast cancer cell line and gene expression on HIF1α. These findings suggest that relevant compounds could be potential therapeutic options for hypoxia-related diseases and cancers that are regulated by the HIF1α pathway. Further studies are required to establish the mechanism of reactions between related drugs and other pathways. Furthermore, these findings should be evaluated with in vivo animal models, which can facilitate and accredit the in vitro results.
Acknowledgments
No Declared
Conflict of interest
No conflict of interest declaration between the authors.
Funding/support
No funding source.
Type of Study: orginal article |
Subject:
Other
References
1. Key TJ, Verkasalo PK, Banks E. Epidemiology of breast cancer. lancet oncol 2001;2(3):133-40. [DOI:10.1016/S1470-2045(00)00254-0] [PMID]
2. Azim HA, Partridge AH. Biology of breast cancer in young women. Breast Cancer Res 2014;16(4):1-9. [DOI:10.1186/s13058-014-0427-5] [PMID] []
3. Favaro E, Lord S, Harris AL, Buffa FM. Gene expression and hypoxia in breast cancer. Genome Med 2011;3:1-12. [DOI:10.1186/gm271] [PMID] []
4. Corcoran SE, O'Neill LA. HIF1α and metabolic reprogramming in inflammation. J Clin Investig 2016;126(10):3699-707.
https://doi.org/10.1172/JCI84431 [DOI:10.1172/jci84431] [PMID] []
5. Li H, Prever L, Hirsch E, Gulluni F. Targeting PI3K/AKT/mTOR signaling pathway in breast cancer. Cancers 2021;13(14):3517. [DOI:10.3390/cancers13143517] [PMID] []
6. Chang D-Y, Ma W-L, Lu Y-S. Role of Alpelisib in the treatment of PIK3CA-mutated breast Cancer: patient selection and clinical perspectives. Ther Clin. Risk Manag 2021:193-207.
https://doi.org/10.2147/TCRM.S251668 [DOI:10.2147/tcrm.s251668] [PMID] []
7. Shanmugam MK, Dai X, Kumar AP, Tan BK, Sethi G, Bishayee A. Ursolic acid in cancer prevention and treatment: molecular targets, pharmacokinetics and clinical studies. Biochem. Pharmacol 2013;85(11):1579-87. [DOI:10.1016/j.bcp.2013.03.006] [PMID]
8. Kashyap D, Tuli HS, Sharma AK. Ursolic acid (UA): A metabolite with promising therapeutic potential. Life Sci 2016;146:201-13. [DOI:10.1016/j.lfs.2016.01.017] [PMID]
9. Semenza G. HIF-1 inhibitors for cancer therapy: from gene expression to drug discovery. Curr Pharm Des 2009;15(33):3839-43. [DOI:10.2174/138161209789649402] [PMID]
10. Goda N, Dozier SJ, Johnson RS. HIF-1 in cell cycle regulation, apoptosis, and tumor progression. Antioxid Redox Signal 2003;5(4):467-73. [DOI:10.1089/152308603768295212] [PMID]
11. Choi YH, Jin GY, Li LC, Yan GH. Inhibition of protein kinase C delta attenuates allergic airway inflammation through suppression of PI3K/Akt/mTOR/HIF-1 alpha/VEGF pathway. PLoS One 2013;8(11):e81773. [DOI:10.1371/journal.pone.0081773] [PMID] []
12. Iqbal J, Abbasi BA, Ahmad R, Mahmood T, Kanwal S, Ali B, et al. Ursolic acid a promising candidate in the therapeutics of breast cancer: Current status and future implications. Biomed Pharmacother 2018;108:752-6. [DOI:10.1016/j.biopha.2018.09.096] [PMID]
13. Shan J, Xuan Y, Zhang Q, Zhu C, Liu Z, Zhang S. Ursolic acid synergistically enhances the therapeutic effects of oxaliplatin in colorectal cancer. Protein Cell 2016;7(8):571-85. [DOI:10.1007/s13238-016-0295-0] [PMID] []
14. Liu R, Chen Y, Liu G, Li C, Song Y, Cao Z, et al. PI3K/AKT pathway as a key link modulates the multidrug resistance of cancers. Cell Death Dis 2020;11(9):797. [DOI:10.1038/s41419-020-02998-6] [PMID] []
15. Book: Kumar AP, et al. Complementary and Alternative Medicines in Prostate Cancer. A Comprehensive Approach. 1 th ed. Boca Raton, FL: CRC Press, Taylor & Francis Group; 2016:93-110. [Google Scholar]
16. Kassi E, Sourlingas T, Spiliotaki M, Papoutsi Z, Pratsinis H, Aligiannis N, et al. Ursolic acid triggers apoptosis and Bcl-2 downregulation in MCF-7 breast cancer cells. Cancer Invest 2009;27(7):723-33. [DOI:10.1080/07357900802672712] [PMID]
17. Gómez Tejeda Zañudo J, Mao P, Alcon C, Kowalski K, Johnson GN, Xu G, et al. Cell Line-Specific Network Models of ER+ Breast Cancer Identify Potential PI3Kα Inhibitor Resistance Mechanisms and Drug Combinations. Cancer Res 2021;81(17):4603-17.
https://doi.org/10.1158/0008-5472.CAN-21-1208 [DOI:10.1158/0008-5472.can-21-1208] [PMID] []
18. Shan J-z, Xuan Y-y, Zheng S, Dong Q, Zhang S-z. Ursolic acid inhibits proliferation and induces apoptosis of HT-29 colon cancer cells by inhibiting the EGFR/MAPK pathway. J Zhejiang Univ Sci B 2009;10:668-74.
https://doi.org/10.1631/jzus.B0920149 [DOI:10.1631/jzus.b0920149] [PMID] []
19. Shan J-z, Xuan Y-y, Zhang Q, Huang J-j. Ursolic acid sensitized colon cancer cells to chemotherapy under hypoxia by inhibiting MDR1 through HIF-1α. J Zhejiang Univ Sci B 2016;17(9):672.
https://doi.org/10.1631/jzus.B1600266 [DOI:10.1631/jzus.b1600266] [PMID] []
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |







 gmail.com
gmail.com